Order taking for Simplera Sync™ sensor and Instinct sensor began in September 2025.
- The Simplera Sync™ sensor began shipping in September 2025.
- The Instinct sensor began shipping in December 2025.

The MiniMedTM 780G system is an automated insulin delivery system designed to reduce highs without increasing lows§ for people living with diabetes.
.png)
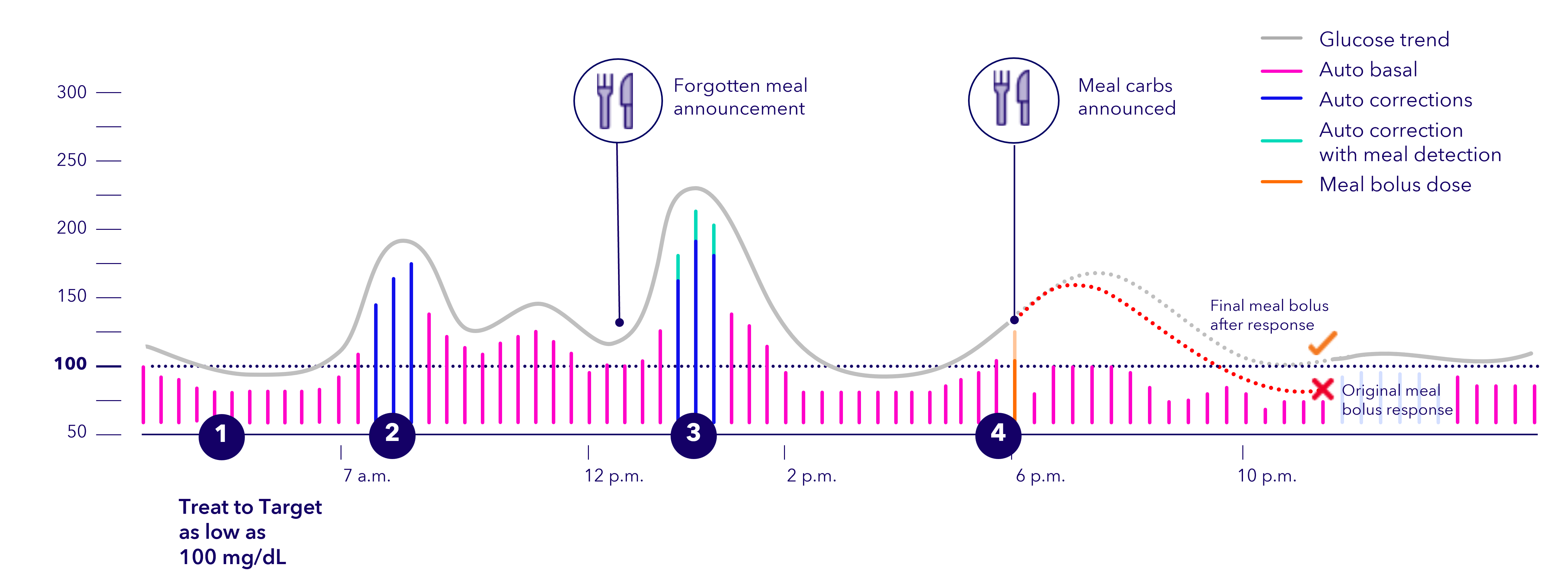
If the system detects a meal based on the sensor glucose's rising rate of change, it can automatically deliver stronger correction doses while sensor glucose values are rising, up to every 5 minutes.
To learn more about how Meal Detection™ technology† works, watch this video featuring distinguished engineer, Lou Lintereur.
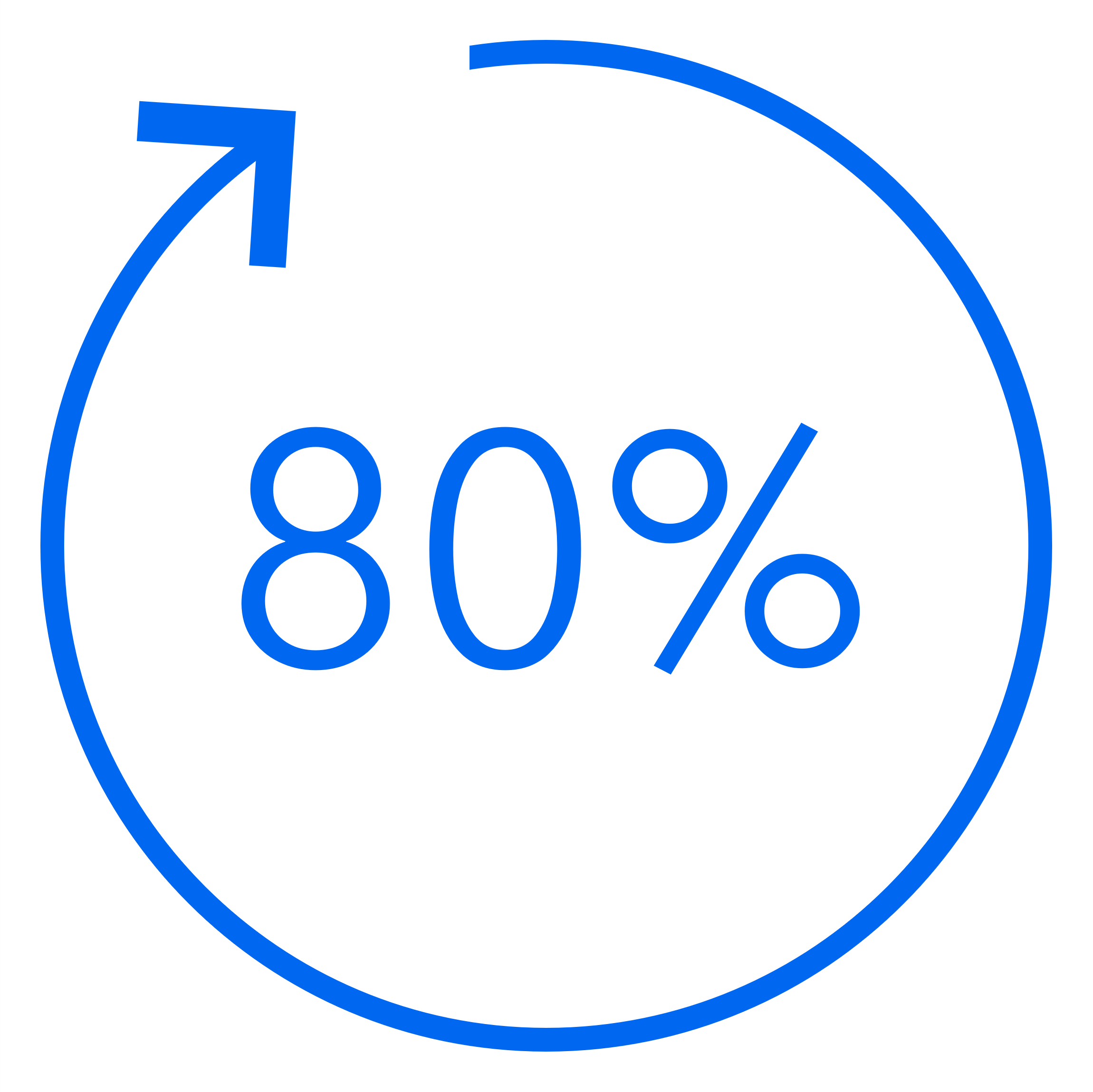
80% Time in Range seen worldwide¶
Recommended settings¶ of 100 mg/dL and 2 hr AIT have shown consistent results globally.2 Real-world results# in adults showed patients achieved up to 80% Time in Range with recommended settings¶ without increasing hypoglycemia.2
Compatible sensors
Medtronic Extended™ infusion set
With only one set change per week, they can experience 96% fewer injections compared to multiple daily injections.◊
Meal Detection™ technology
Automatically anticipates and corrects blood glucose every 5 minutes‡ even when carb counting isn't exact or an occasional bolus is missed.§


79.8% Time in Range achieved for people with T2D despite 63% of participants in the T2D pivotal trial not having prior technology experience including either an insulin pump or CGM system.6
MiniMedTM 780G - ADAPT Study
Download
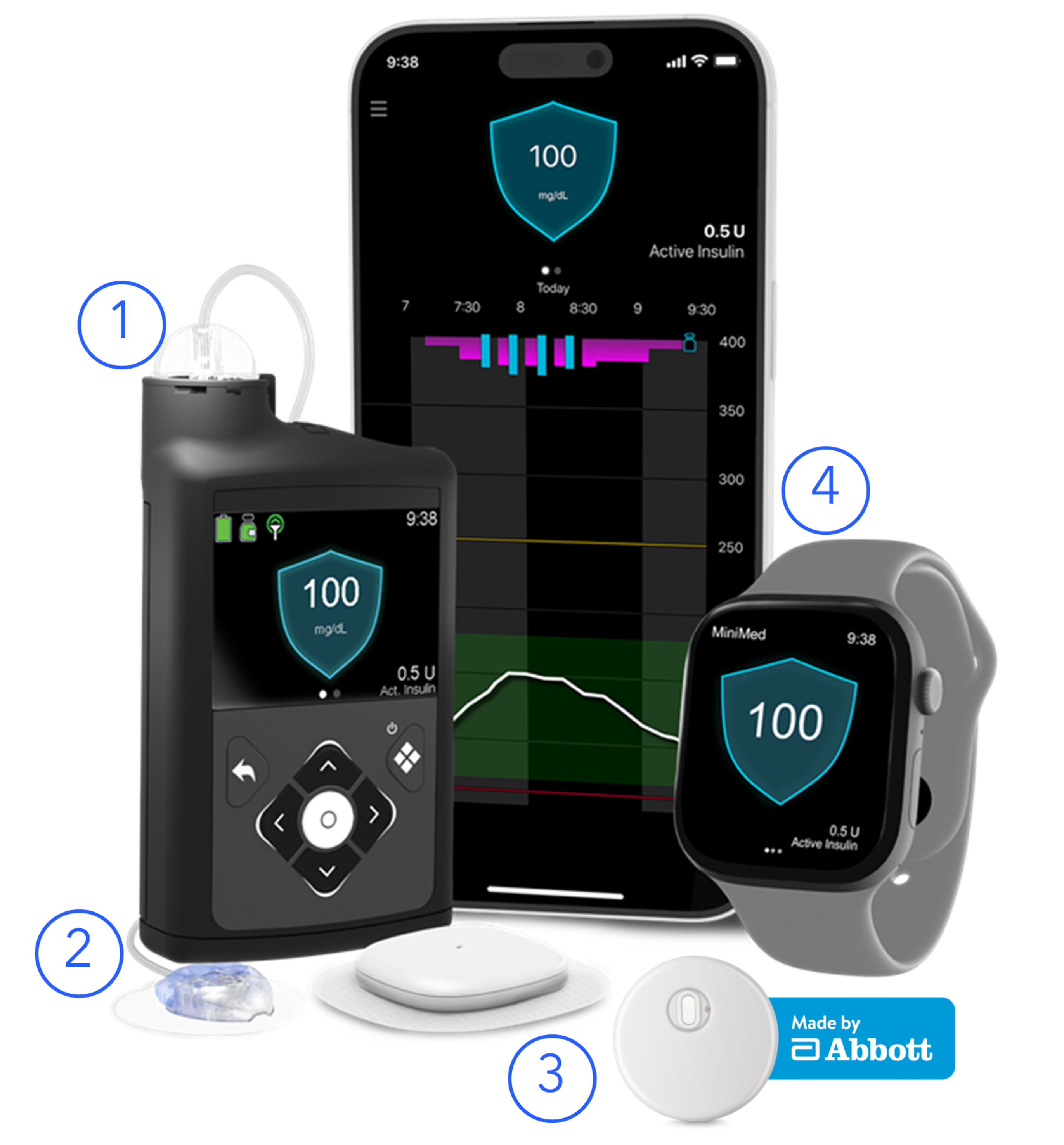
1. MiniMedTM 780G insulin pump
with advanced SmartGuardTM technology
2. Medtronic ExtendedTM infusion set
for up to 7 days of wear
3. Compatible sensors
Instinct sensor: Proven to be accurate, stable, and consistent for 15 full days, without fingersticks.∞4,5
Simplera Sync sensor: All-in-one sensor with easy insertion that measures glucose levels every 5 minutes for up to 7 days.
4. MiniMedTM Mobile app & Apple Smart Watch® compatibility
Allows patients to view glucose levels, pump information, and insulin data on their phone or Apple Watch. View this reference chart for a list of compatible smartphones.
CareLinkTM Clinic
CareLinkTM Clinic reports are the best-in-class for treatment of patients on automated insulin delivery (AID) therapy.∆
The easy-to-use sensor, infusion set, 300u reservoir, and insulin pump are designed to work together seamlessly. With only one set change per week, they can experience 96% fewer injections compared to multiple daily injections.◊
When will the new sensors for the MiniMed™ 780G system begin shipping?
Order taking for Simplera Sync™ sensor and Instinct sensor began in September 2025.
Who is this MiniMed™ 780G system recommended for?
The MiniMed™ 780G system is approved for people with type 1 diabetes ages 7 and above and adults ages 18 and above living with insulin-requiring type 2 diabetes.
What are the recommended settings for the MiniMed™ 780G system?
The MiniMed™ 780G system safely delivers insulin without increasing hypoglycemia when using recommended settings¶ of 100 mg/dL and 2-hour Active Insulin Time (AIT).2
How do I prescribe the MiniMedTM 780G system?
Yes, a new prescription is required for the MiniMed™ 780G system. This applies to the software upgrade as well.
MiniMed™ Distribution Corp.
NCPDP ID: 5900990
NPI number: 1831418672
18302 Talavera Ridge
San Antonio, TX 78257-2605
Phone: 877-891-6750
In the direction section of the ePrescribe screen or on written prescriptions:
MiniMed™ 780G pump
NDC: 63000-075854
Include the following:
Extended infusion set & Reservoir
NDCs: 05244-0023-28 & 63000-0497-18
Include the following:
Instinct™ sensor
NDC: 57599-0880-00
Simplera Sync™ sensor
NDCs: 63000-056504
Guardian™ 4 sensor & transmitter
NDCs: 63000-0445-15 & 63000-0413-38
Indicate the following:
If we need any additional information, we will contact your office.
Has Medicare approved the MiniMed™ 780G system?
Yes, the FDA has approved the MiniMed™ 780G system and Medicare has completed the coding verification process. Patient orders will follow the normal processes for shipping.
Do patients need to do fingersticks when using the Simplera Sync™ or Guardian™ 4 sensor?
Using the system does not completely replace fingersticks but patients will likely check their blood glucose less frequently. The Simplera Sync™ sensor and Guardian™ 4 sensor do not require a blood glucose fingerstick to calibrate the system or make treatment decisions with SmartGuard™ technology†. For much of the time, patients will not need to do a fingerstick. However, when using the Simplera Sync™ or Guardian™ 4 sensor with the MiniMed™ 780G system, safety precautions were put in place to maximize the performance of the SmartGuard™ algorithm. Because of this, there are instances when the pump will prompt a blood glucose request from the user.
* Refers to SmartGuard™ technology. Some user interaction required. Individual results may vary.
† Fingersticks required in manual mode & to enter SmartGuard™. If symptoms don’t match alerts & readings, use a fingerstick. Refer to user guide. Pivotal trial participants spend avg of > 93% in SmartGuard™.
Is the MiniMed™ 780G insulin pump waterproof?
At the time of manufacture and when the reservoir and tubing are properly inserted, your pump is waterproof. It is protected against the effects of being underwater to a depth of up to 12 feet (3.6 meters) for up to 24 hours. This is classified as IPX8 rating. See user guide for more details. The sensor is water-resistant up to 3 feet (1 meter) for up to 30 minutes. Do not immerse the sensor for longer than 30 minutes. CGM readings may not be transmitted from the CGM to the pump while in water.
What services are offered to people who are interested in obtaining this system?
Access programs
We help to find the lowest out-of-pocket cost option by offering a variety of access and affordability programs based on insurance coverage.
Comprehensive insurance support
Our team verifies insurance, collects and submits prior-authorization documents if needed, and checks pharmacy benefits.
Support and training
Helping you train your patients online or face-to-face; a 24/7 helpline can help them tackle their daily challenges.
Delivery plan
Automatic recurring diabetes supply shipments delivered when and where your patients need them.
Device upgrades
Whether they’re in or out of warranty, customers may receive up to $500 credit for trading in an old pump when upgrading to new device.
† Taking a bolus 15 – 20 min before a meal helps to keep blood sugar levels under control after eating.
‡ Refers to auto correct, which provides bolus assistance. Can deliver all correction doses automatically without user interaction, feature can be turned on and off.
§ Refers to SmartGuard™ technology. Individual results may vary.
◊ Assumes 4 injections per day for 30 days and one infusion set change every seven days.
¶ Recommended settings in adults: SmartGuard™ Target: 100 mg/dL, 2 hr AIT, ages <=15: Start at SmartGuard™ Target: 110mg/Dl and reduce to 100 mg/Dl if no hypos, 2 hr AIT
# Due to inherent real-world study limitations, caution is advised when attempting to extrapolate these results to new patients. There could be significant differences.
∆ Medtronic Diabetes: CareLink™ Clinic Market Research Survey, n=379; Q1 2025: P. 9-10 (February 2025)
∞ Fingersticks required in manual mode & to enter smartguard. If symptoms don’t match alerts & readings, use a fingerstick. Refer to user guide. Pivotal trial participants spend avg of > 93% in SmartGuard.
1. Medtronic data on file: MiniMed™ 780G users survey conducted in April – May 2021 in UK, Sweden, Italy, Netherlands and Belgium. N = 789 †comparison of MM670G/MM770G pivotal with MiniMed™ 780G system w/G4S CAS (Avg. 12.12 for MM670G and 7.61 for MiniMed™ 780G system in a day)
2. Arrieta A, et al. Diabetes Obes Metab. 2022;10.1111/dom.14714.|
3. Choudhary P, et al. Lancet Diabetes Endocrinol. 2022;10(10):720-731.
4. Data on file, Abbott Diabetes Care, Inc.
5. Finger pricks are required if glucose readings and alarms do not match symptoms or expectations.
6. Bhargava A, Bergenstal RM, Warren ML, et al. Safety and Effectiveness of MiniMedTM 780G Advanced Hybrid Closed-Loop Insulin Intensification in Adults with Insulin-Requiring Type 2 Diabetes. Diabetes Technol Ther. 2025;27(5):366-375. doi:10.1089/dia.2024.0586
Important Safety Information: MiniMedTM 780G system with SmartGuardTM technology with Instinct sensor, Simplera SyncTM sensor, and GuardianTM 4 sensor
The MiniMedTM 780G system is intended for the continuous delivery of basal insulin at selectable rates and the administration of insulin boluses at selectable rates for the management of type 1 diabetes mellitus in persons 7 years of age and older, and of type 2 diabetes mellitus in persons 18 years of age and older requiring insulin. The system is also intended to continuously monitor glucose vales in the fluid under the skin.
The MiniMedTM 780G System includes SmartGuardTM technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values. The system is intended for use with connected sensors, including the Simplera SyncTM and GuardianTM 4 sensors and integrated continuous glucose monitors, including the Instinct sensor, each of which has different wear-time, form factor, insertion site, and other distinguishing characteristics that relate to sensor performance. Consult the appropriate sensor user guide when using the system. Discuss treatment decisions with your HCP.
WARNING: Do not use the SmartGuardTM feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuardTM feature.
WARNING: Do not use MiniMedTM 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of MiniMedTM 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuardTM feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a blood glucose (BG) meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMedTM 780G system has not been studied in pregnant women or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including user guides and important safety information such as indications, contraindications, warnings and precautions associated with system and its components, see https://bit.ly/MiniMedRisks.
©2026 MiniMed. MiniMed and MiniMed logo are trademarks of Medtronic MiniMed, Inc. The sensor shape and appearance, Abbott, and “a” logo are marks and/or designs of the Abbott group of companies in various territories and used under license. Sensor image ©2026 Abbott. TM*Third–party brands are trademarks of their respective owners.
DreaMed Diabetes is a trademark of DreaMed Diabetes, Ltd. The MiniMedTM 780G system algorithm includes technology developed by DreaMed Diabetes.